Drug Evaluation
The implementation of our CE-MS technology is possible from the very beginning of preclinical development (animal models) to the point of phase III or rather phase IV clinical trials. We are highly experienced in the efficient and time-saving assessment of newly developed as well as already existing pharmaceutical substances. Our approach in clinical proteomics enables the accurate clinical characterisation of patients and controls (high fidelity phenotyping). CE-MS profiling will accelerate drug development by ascertainment of statistically significant data from a small number of patients (defined cohorts) plus an enhancement in the determination of pharmaceutical drug safety.
Disease state specific polypeptide patterns obtained by our CE-MS technology help to select an appropriate therapy. Therapeutic effects of administered drug can be displayed and controlled in the polypetide patterns. This enables optimization of applied therapy (e.g. dosage modification).
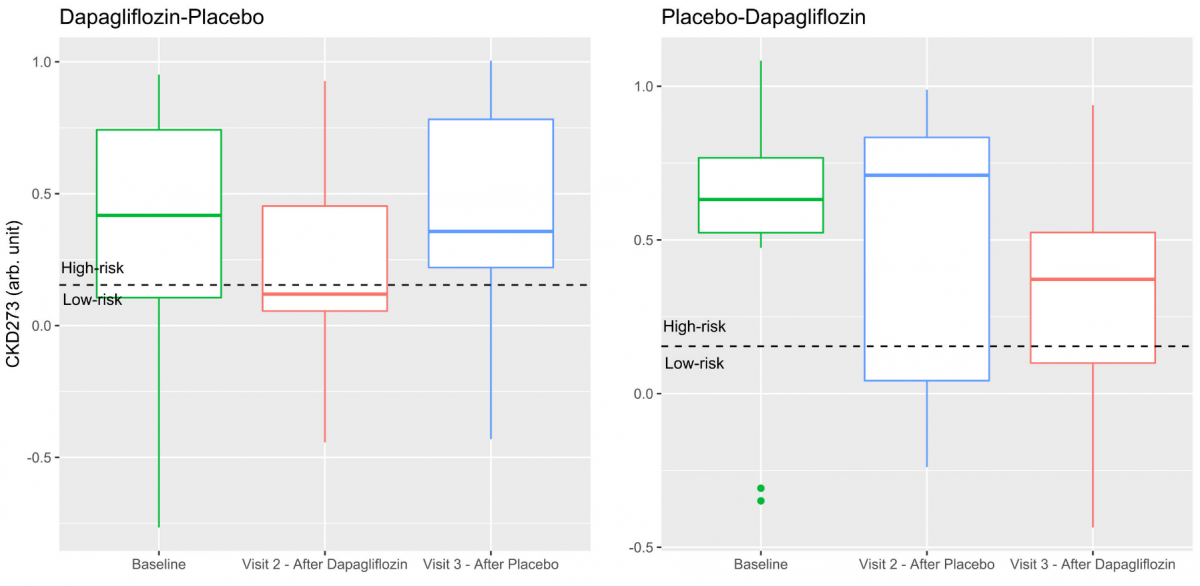
Figure 1: Boxplots illustrating the median (horizontal line), inter-quartile range (boxes), and min/max values (whiskers) of the CKD273 classifier score at baseline, after dapagliflozin treatment, and after placebo. The left panel illustrates the distribution of the participants treated with the sequence dapagliflozin-placebo, and the right panel the participants treated with the sequence placebo-dapagliflozin [see Curovic et al.].
For more information, please click on the following links:
MOS benefits in drug development (PDF)
MOS unique seling proposition (PDF)
REFERENCES
- Curovic VR, et al. Dapagliflozin Improves the Urinary Proteomic Kidney-Risk Classifier CKD273 in Type 2 Diabetes with Albuminuria: A Randomized Clinical Trial. Diabetes Care 2022, 45(11):2662-2668.
- Latosinska A, et al. SGLT2-Inhibition reverts urinary peptide changes associated with severe COVID-19: An in-silico proof-of-principle of proteomics-based drug repurposing. Proteomics 2021, 21(20):e2100160.
- Mokou M, et al. A Novel Pipeline for Drug Repurposing for Bladder Cancer Based on Patients' Omics Signatures. Cancers (Basel) 2020, 12(12):3519.
- Siwy J, et al. Urinary Proteomics as a Tool to Identify Kidney Responders to Dipeptidyl Peptidase-4 Inhibition: A Hypothesis-Generating Analysis from the MARLINA-T2D Trial. Proteomics Clin Appl. 2019, 13(2):e1800144.



